Our Group Companies
Our company is promoted by a team of experienced, technically competent & industrially knowledge personalities in the area of Biotechnology, Biochemistry, Phytochemistry, Pharmaceuticals, mechanical engineering, automation engineering, electrical and instrumentation engineering and Process Engineering & Agriculture.
All our process technologies are based on intense research and development. We also do new process development according to the client’s needs. We are supported by a strong fabrication facility which helps in fabricating and installing the process equipments and machineries. This fabrication facility is supported by mechanical engineers, CADD specialists’ instrumentation and piping engineers and welders and other resources.
All our turnkey supplies conform to USFDA norms of design qualification (DQ) protocols, installation qualification (IQ) protocols and operation qualification (OQ) protocols.

Our Quality Policy
In Clean Green Biosystems, all procedures and work process conform to strict international industrial standards
“Our projects, services and products are developed in a safe, secure & environment friendly manner with personal commitment of PDCA (Plan-Do-Check-Act) manner to understand, meet, and fulfill our client’s requirement, as well through continual improvement making Clean Green Biosystems as truly a Green Global Company with values.
“We are dedicated to deliver our projects on-time with eco-friendly and economic technology”.
Clean Green Biosystems is now accredited with International standards of ISO 9001:2015 (Quality Management System), and planning to obtain ISO 14001 (Environment Management System), OHSAS (OCCUPATIONAL HEALTH & SAFETY ADVISORY SERVICES) & ISO 27001 (Information Security Management System).
The specialty of our equipments and Plant and machineries are:
All our equipments are made with food/Pharma grade SS materials.
We provide Zero bacteria valves.
Our piping work conforms to food/Pharma grade.
Our plants are always versatile in nature.
Our plants are energy efficient.
We provide skid mounted plant and machineries.
We offer cGMP plants.
Our process technology involves low temperature short contact distillation path.
Clean Green Biosystems – System USP
Our System Throughput
Processing system to process 25 kgs/hour to 20 TPD of fresh / dry biomass
Liquid-Liquid extraction system to process 25 Liters to 20 KL
Distillation and concentration system (integrated solvent recovery system) to process 25 LPH to 5 KLPH
Customized capacities as per client’s needs
In-house developed and commercially validated process engineering software's used
AutoCAD and Solid works software's used to engineer the equipment’s and assemble the equipment’s into a system
Our System Safety Compliance
SS-316, ASME complaint equipments
Welding of the pressure parts as per international standards
FLP electrical components
Closed loop solvent recovery
Low temperature and no pressure operations
Equipments inspection CSA, SGS, UL, CE listed
Our Safety Versatility
Can use multiple solvents
Can use both hot extraction and cold extraction
Can be used for multiple products
User Friendly Operation
Detailed SOP
PLC-SCADA based automation
HMI screen at every process station
Pre-programmed process for customized and standard operations
Skid mounted / pre-installed equipments
Plug and start small scale systems
Project Deliverables and Time Frames
Month 1 – Confirmation
Order Confirmation
Reconfirmation of design
Fabrication drawing completion, pending client approval
Beginning of fabrication work
Order placement for bought out items.
Month 2 – Inspection
Continuation of fabrication work
Site condition inspections
First inspection of fabricated equipments
Month 3 – Site Evaluation
Second inspection of fabricated equipment
Receipt of bought out items
Dispatch of bought out items to installation site
Starting erection of structural skid at site.
Month 4 – Site Erection
Dispatch of fabricated items to the installation site against pro forma invoice
Starting site erection of equipment
Month 5 – Installation
Continuing the site work of equipment installation, piping and instrumentation work.
Month 6 – Optimization
Completion of installation
Trail run and optimization
Commissioning and handing over the plant.
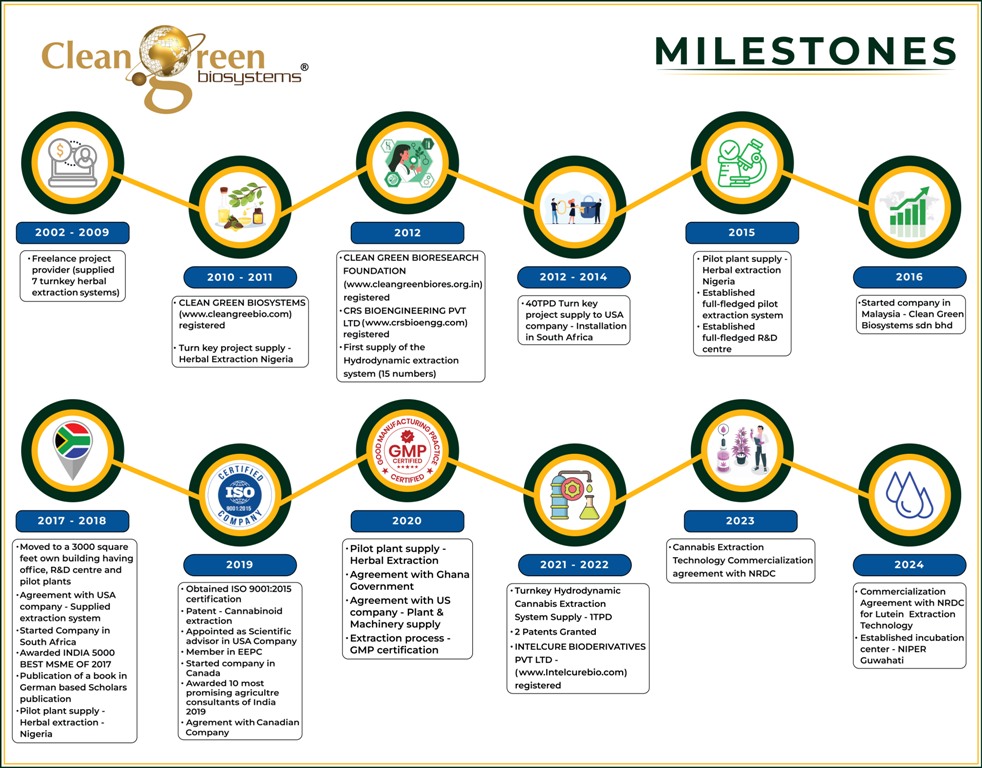
Our Growth